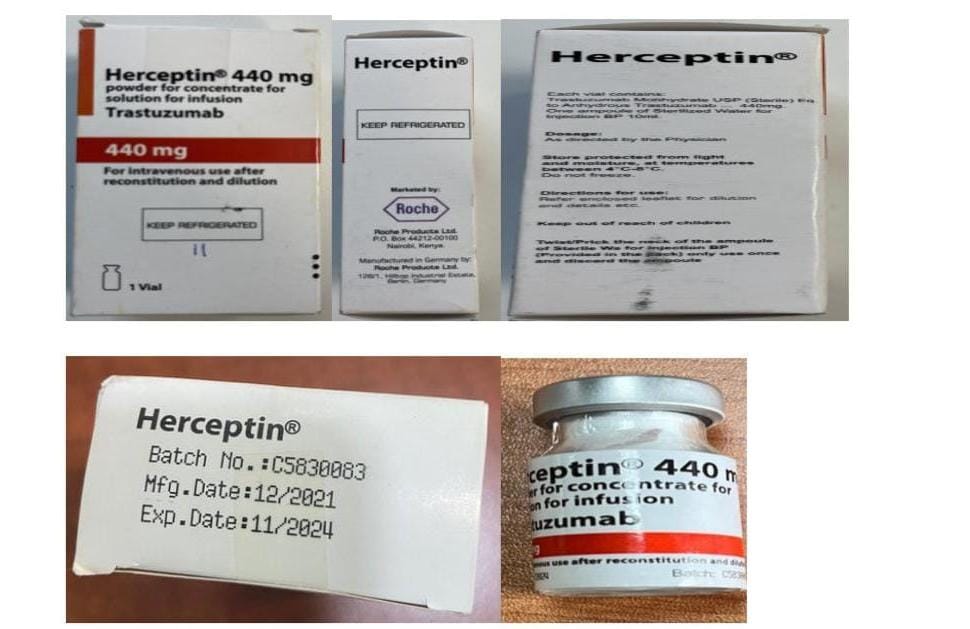
Poisons board issues warning on counterfeit Herceptin 440mg detected in Kenyan market

The Pharmacy and Poisons Board (PPB) has issued an alert over a suspicious batch of counterfeit breast cancer treatment drug, Herceptin 440mg (Trastuzumab 440mg), detected in the market.
According to the board, the product batch is claimed to be manufactured in Germany by Roche Products Limited, bearing the Batch number C5830083, manufactured in December 2021 and expected to expire in November 2024.

“The public is notified that this specific product batch number C5830083 is a confirmed counterfeit. It is not authorised to be in the market and is a falsified product in view of the falsified contents, packaging and labelling aspects which have been confirmed by the brand owner,” warned the board.
The board cautioned the public against the use, supply, sale or distribution of the product batch number C5830083 stating that the safety, quality and efficacy cannot be assured.
The board, in collaboration with government investigative agencies, has instituted legal and regulatory actions against individuals suspected to be perpetrating the illegal activities in contravention of the Pharmacy and Poisons Act (Cap. 244).
“The PPB has initiated a rapid response and heightened surveillance of the market to ensure that safety and public health is protected. We would like to assure the public that PPB has established robust market surveillance and control system that continuously monitor the quality and safety of medical products in the Kenyan market.”
The board is urging members of the public and healthcare providers to report and volunteer information on suspicious activities that may be related falsification, as well as suspicious, substandard, and falsified medical products through its official communication channels.

